Jim Palandri
Research Associate
Department of Earth Sciences
1272 University of Oregon
Eugene, Oregon 97403-1272
Jim might be found in:
- Ilya
Bindeman's stable isotope lab
- Elsewhere in Cascade or Columbia fixing something
- TSA's various shops -
the folks who can build almost anything
An ongoing project since 2022 is restoration
of Gastherm.dat,
the thermodynamic database for GASWORK, SOLVGAS, and VOLCAL. These
volcanic gas modeling programs are analogous to Chim-XPT,
Solveq-XPT, and Geocal-XPT except that the mobile phase carrying all
of the chemical components is gaseous rather than aqueous. Related items
Handy links to mostly local reference info and where off-site* may
be broken
Items related to local applications Chim-XPT, Solveq-XPT, and
Geocal-XPT —
- Soltherm2023
| Solprint
— Latest version derived from mineral & gas data in tc-ds636,
still citing Holland and Powell (H&P) 2011
- Soltherm2011
| Solprint
— Previous version derived from tc-ds62
citing H&P 2011.
Here adding tourmaline and basis set component #66†
univalent lithium. Sweet, sweet, lithium
- Solveq & Chim-xpt demo input files
for both of the above, with very slightly different output
- Gas stoichiometry data for Geocal-XPT — Gas.sto
| Reactant stoichiometry data for Chim-XPT — Minox.dat
- SolthermBRGM.XPT
| SolprintBRGM.txt
— *BRGM *Thermoddem data,
reformatted from the ToughReact *version.
For the Martians, and other low temperature peoples
- XPT-LogK
— a utility for extracting and interpolating Soltherm log K's
- Manuals — Solveq-XPT
and Chim-XPT
- Fugacity coefficients in mixtures of H2O, CO2,
and NaCl at high P and T: program FUGCO,
with a worked example
as in the cited reference, and included as subroutines in Chim-XPT
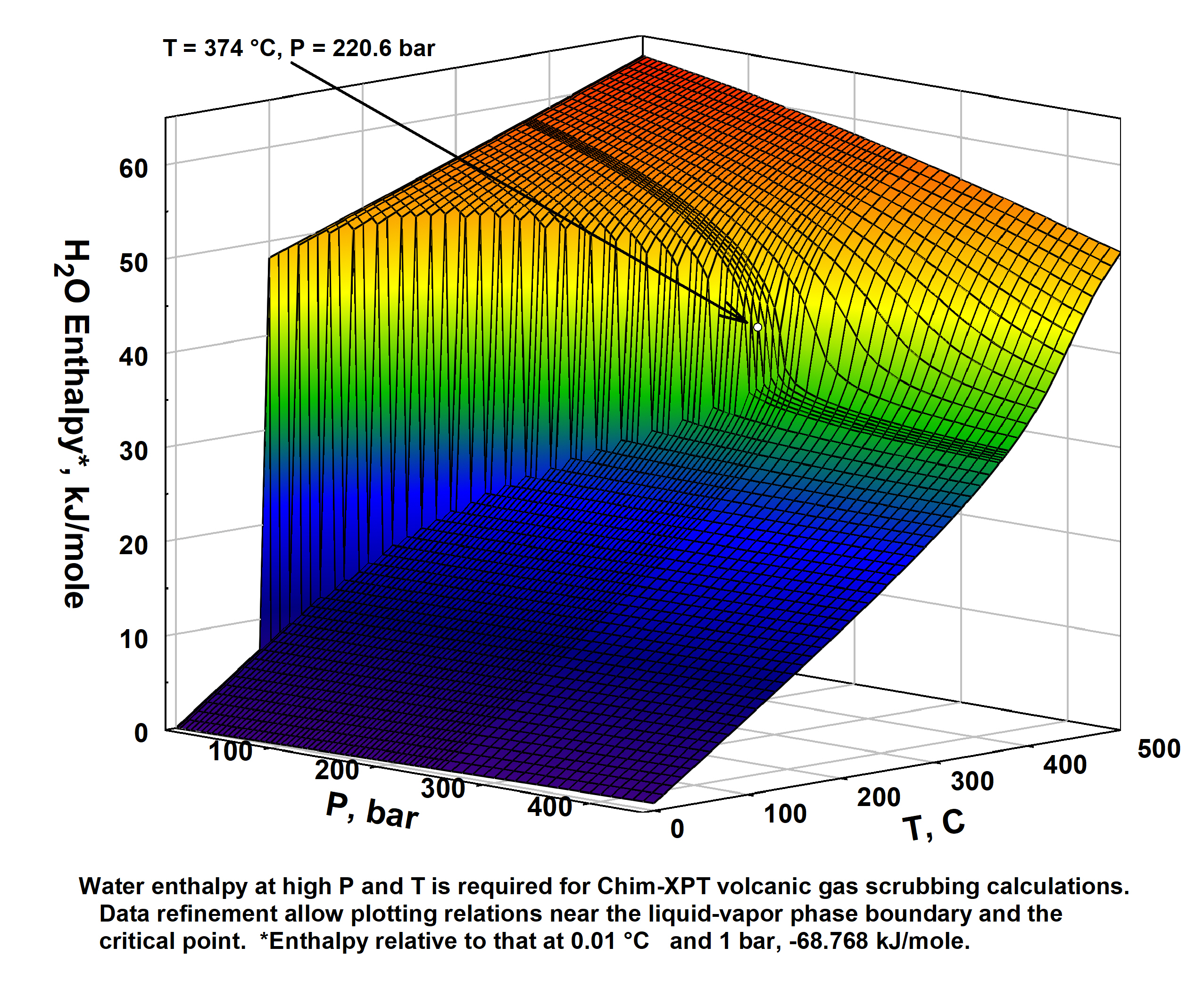
- Delicious water, what are thine properties?
So too ethereal vapor phase thereof - Steam Tables - pdf,
csv
Supcrt-related items for computing log K's in Soltherm —
- Supcrt92.zip
— the original distribution from Johnson et al. 1992
using Maier-Kelly heat capacity, Cp = a + b T + c T-2
- Supcrt96 | Source
| Win32
| x64
— derived from Supcrt92 to use Maier-Kelly heat capacity, and adding
Powell and Holland 1985,
Cp = a + b T + c T-2 + d T-0.5
- Cprons96 | Source
| x64
— derived from Cprons92. Converts any (including any of those for
Supcrt92) properly formatted sequential access database to direct
access, suitable for use with Supcrt96
Some items related to the data used by Supcrt —
- *HPx-eos
& Thermocalc
— for phase equilibria computations — application and data from Tim
*Holland.
The primary application for which the H&P 2011
compiled their data
- The most current Supcrt92 data might be found at the *Gitlab ENKI portal, in Everett
Shock's *Geopig
workgroup space, file *Slop16.dat.
This group has done phenomenal work in adding data for organic
compounds.
Some info about files SPRONS and DPRONS — Sequential-, and Direct-access
PROperties of Natural Substances, as per
Johnson et al 1992.
The sequential access file is for data editing, which is processed
by Cprons into the direct access version for increased Supcrt
execution speed
- SPRONS92.DAT
— The original masterpiece
- Sprons96_HP2023.dat
— Uses the most current aqueous species data from *Slop16.dat,
internally consistent mineral and gas data from the H&P *Thermocalc
web site (file tc-ds636.txt,
2023), and additional data that complement them
- Dprons96_HP2023.dat
— The above file piped through Cprons96 and ready to use with
Supcrt96
- Sprons96_HP2011.dat
— The previous version using data from *Slop07.dat,
and earlier H&P data (file tc-ds62.txt,
2011)
- Some background info
on the H&P data
- Sprons96-xpart-HP2023.dat
— The most recent and still citing H&P 2011
- Sprons96-xpart-HP2011.dat
— Previous version
- Sprons96-xpart-gases.dat
— The gases section, expanded to 758 items for the GASTHERM project
- Sprons96-xpart-misc.dat
— Bonus mineral and gas phase data from other sources, also properly
formatted for Supcrt96. Many of these are from Slop16.dat
- Sprons92-xpart-aqs.dat
— Bonus aqueous phase species data too, properly formatted for any
version of Supcrt. In particular, H4SiO4,aq.
from Stefánsson 2001;
rarely does so very much hinge upon so very little
Miscellaneous links:
Experimental petrology —
- CSPV North
— LabVIEW v.2012sp1/WinXP
- CSPV South
— LabVIEW v.2018/Win10-64
- Related items
Isotope ratio mass spectrometry —
- Info on laser fluorination
and extraction of oxygen from earth materials, for d18O and d17O
isotopic analysis
- Reference and other info. Lots of
photos, mostly to help in putting Humpty together again in semblance
of working order
Fluid inclusions —
- Some photos
of the setup, before it was boxed up
Just for fun —
- Geologic map [A,
B,
L;
USGS 7.5' - Clarno,
Muddy
Ranch] of Muddy Ranch and vicinity, north of Currant and Muddy
Creeks, Wasco, Wheeler, and Jefferson Counties, Oregon (1993). Aka Washington
Family Ranch, and before that Rajneeshpuram
- Streetcar map
of Eugene, OR, 1907-1928 — Exposed track is still be seen on Moss,
Columbia, and University Streets. Modified from a map
published in Eugene Weekly in 2008
- Some photographs
of the I-5 Whilamut
Passage Bridge replacement and related construction, near
milepost OR 192
- Related items
† Bringing us to 55 of 82‡
stable elements in the Soltherm basis set. The other items being
metastable acetate, oxalate, succinate, malonate, aqueous CH4,
H2, N2, and NH3, radioactive UO2+
and Pm+3, and alternate redox component O2 -
redox state is defined by either paired sulfate-sulfide or H2O-O2.
A legacy from the era of 16-bit operating systems, when use of
sulfide for an oxidizing system, or O2 for a reducing
system, might cause an array underflow — or that was claimed to be
the case but will never be verified. Since water must be present and
sulfate is so common, basis components sulfide and O2
were hard coded to be mutually exclusive. Pick one.
‡ Bismuth-209 undergoes α-decay to 205Tl with
a half-life of 2.01x1019 years, or, ~1.46x1009
times the 1.38*1010 or so year age of our observable
universe. To not be a pedant but only in this one case, bismuth is
deemed stable. So 82 rather than 81.
Drumknott!
- Vetinari