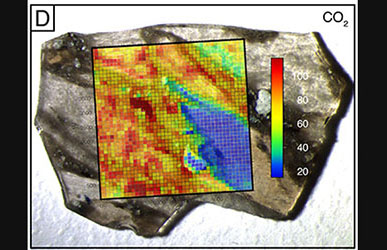
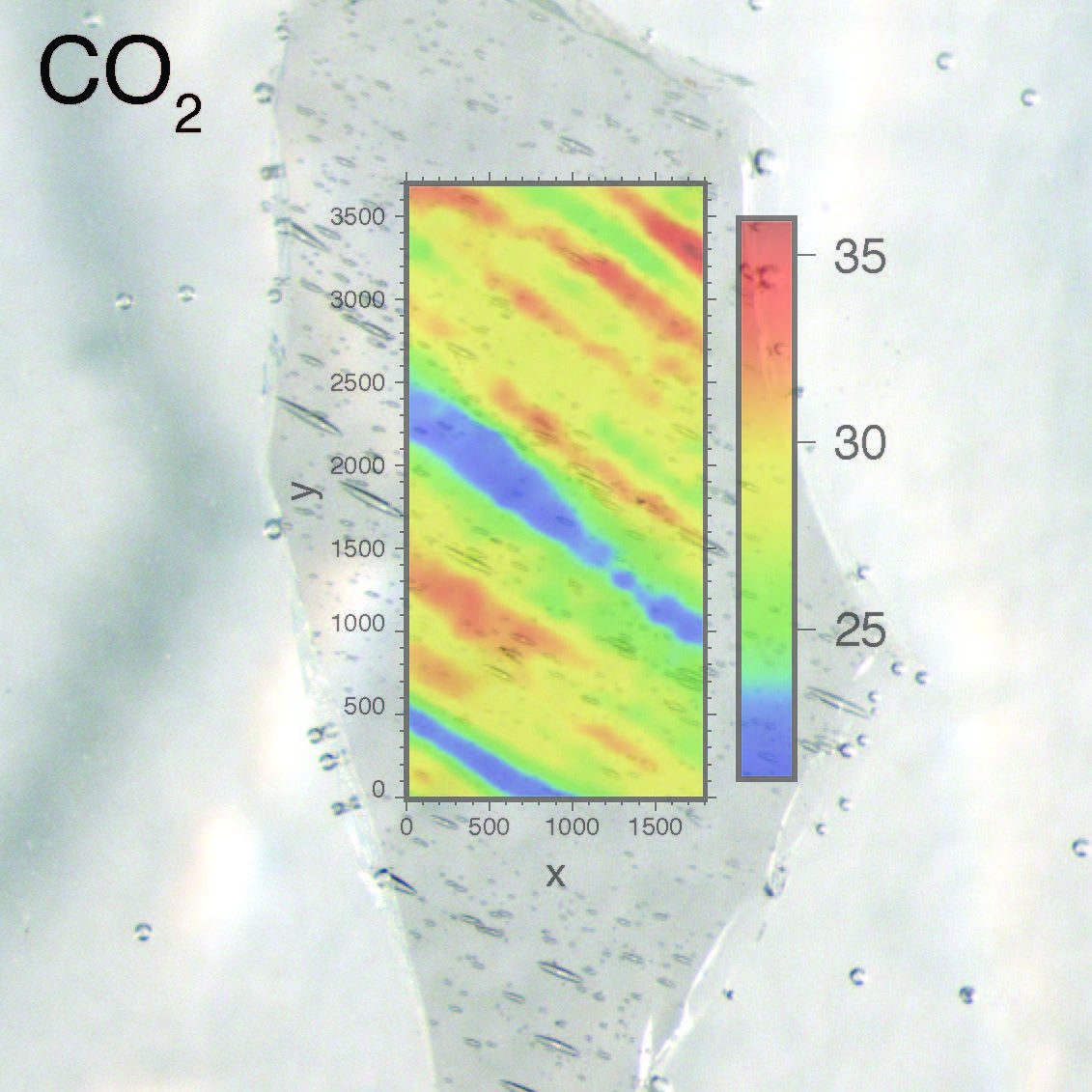
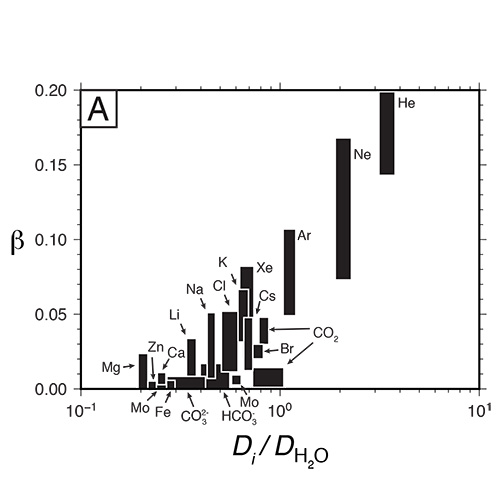
If you're trying to decide which graduate school you'd like to attend, here are some reasons why you should choose the University of Oregon in Eugene.
Experimental geochemistry @ UO
I'm looking for graduate students to join my group. If you're interested in attending graduate school at UO please send me an email with a copy of your CV and a statement of why you're interested in working with me. Our graduate student applicant pool is very competitive and I'm particularly interested in taking students that have ideas of what they'd like to work on and why we'd be a good fit. There are currently no funded postdoctoral positions availale but if you're interested in writing a proposal to come to Oregon please let me know.
DoES @ UO
We're a big department at 20 faculty members and 40 graduate students, but we're not too big. You'll still get regular attention from your advisor, have the chance to interact with all faculty members, and there's a healthy graduate community to support you in your research endeavors. Many of our recent graduates have gone on to prestigious postdocs at national institutions, such as the USGS, or to other top rated Earth science programs while others acquire faculty positions at schools throughout the US.
Eugene
Eugene is a big-small town. You can find world class fly fishing along the Willamette river, rafting swimming or tubing on the McKenzie, golf in Eugene (Laurelwood) or any of the fine courses nearby (I like Diamond Woods and Tokatee). There are local farmers markets in Eugene on the weekends or you can go to the farm if you prefer. Oregon beer is legendary. You'll have the opportunity to choose from a dozen local breweries in town, or if beer doesn't suit your fancy, the Willamette Valley is up and coming a Pinot noir powerhouse, with several wineries within a few miles of town. For a good time, you can also check out the Oregon Country Fair, the Bach Festival, and Saturday market.
Oregon
An hour drive west of Eugene, you'll find the breathtaking Oregon coast where you can dive, fish, surf, and eat local, all in a day trip. An hour to the east are the Cascades. You can see the Three Sisters volcanoes from Eugene and both downhill and cross country skiing are slightly over an hour away. Two hours north, you'll find Portland, OR. A beer, dub-step, and hipster mecca, you'll need to make a pilgrimage north at some point, if not for Portland itself, to ski above the clouds at Mt. Hood or tour the spectacular waterfalls of the Columbia River Gorge. Two plus hours south is Crater Lake National Park. You'll have your choice of stratovolcanoes, from Mt. Shasta in northern California, to Mt. St. Helens just north of the Oregon-Washinton border....there's plenty to climb and study. The Rogue, McKenzie, and Umpqua rivers are also fun to raft and most are available as day trips. If you like to rock climb, you'll find no shortage of places to do so (Smith rock, near Bend, OR, is well known as the place where sport climbing was invented).